The Freundlich equation or Freundlich adsorption isotherm, an adsorption isotherm, is an empirical relation between the concentration of a solute on the surface of an adsorbent to the concentration of the solute in the liquid with which it is in contact. In 1909, Herbert Freundlich gave an expression representing the isothermal variation of adsorption of a quantity of gas adsorbed by unit mass of solid adsorbent with pressure.[1] This equation is known as Freundlich adsorption isotherm or Freundlich adsorption equation. As this relationship is entirely empirical, in the case where adsorption behavior can be properly fit by isotherms with a theoretical basis, it is usually appropriate to use such isotherms instead (see for example the Langmuir and BET adsorption theories).
#SurfaceChemistry
ABOUT CHANDRAHAS SIR:-…………………………………….
S&K CLASSES is the first choice of student in Northen India for preparation of JEE main and advanced ,NEET & AIIMS.
Chandrahas Sir is first online teacher of chemistry in Dehradun.
Chandrahas Sir is known for his focused and simplified JEE main and advanced/NEET/AIIMS/BOARD teaching to bring to student an easy and analytical methodology towards JEE main and advanced / AIIMS.
* Online webinar for chemistry -https://www.youtube.com/edit?o=U&video_id=n_gbVJj5PLs
*solid state | position of octahedral &tetrahedral voids in F.C.C-https://www.youtube.com/edit?o=U&video_id=6jrGVWVRXMU
*How to memorize 7 crystal system -https://www.youtube.com/edit?o=U&video_id=kc7DaJccjFs
Please do let me know if you have any doubt about this chapter
I APPRECIATE YOUR VISIT TO MY CHANNEL. THANK YOU
source













![CY_GATE_2019_PHYSICAL_SPECTROSCOPY_[ELECTRONIC_BASIC]_All IN ONE_[Short_Trick]_2018-19_PART_1ST - Videos](https://trends.edugorilla.com/wp-content/uploads/sites/8/2018/08/cy_gate_2019_physical_spectroscopy_electronic_basic_all-in-one_short_trick_2018-19_part_1st-218x150.jpg)








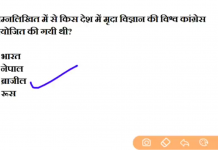






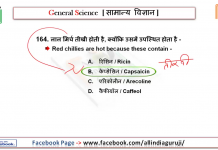


![24 August 2018 – The Indian Express Newspaper Analysis हिंदी में – [UPSC/SSC/IBPS] Current affairs - Videos](https://trends.edugorilla.com/wp-content/uploads/sites/8/2018/08/a520-218x150.png)




sir plzz physical chemistry numericals tricks plzz plzz maine kai baar aapko request kiys he plzz start with equilibrium and thermodynamics tricks plzz plzz plzz
Nice video sir