Concentrated nitric acid used in laboratory work is 68% nitric acid by mass in aqueous solution. What should be the molarity of such a sample of the acid if the density of the solution is 1.504 g mL–1?
Our Official Facebook Page : https://www.facebook.com/livetutelage
Our Official Youtube Channel : https://www.youtube.com/c/LiveTutelage
Online Test Preperation : http://livetutelage.com/test-preparation/
Online Library : http://livetutelage.com/library/
source













![CY_GATE_2019_PHYSICAL_SPECTROSCOPY_[ELECTRONIC_BASIC]_All IN ONE_[Short_Trick]_2018-19_PART_1ST - Videos](https://trends.edugorilla.com/wp-content/uploads/sites/8/2018/08/cy_gate_2019_physical_spectroscopy_electronic_basic_all-in-one_short_trick_2018-19_part_1st-218x150.jpg)








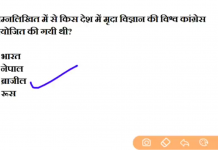






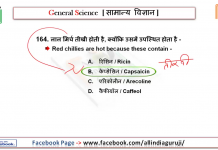


![24 August 2018 – The Indian Express Newspaper Analysis हिंदी में – [UPSC/SSC/IBPS] Current affairs - Videos](https://trends.edugorilla.com/wp-content/uploads/sites/8/2018/08/a520-218x150.png)




Hey, amazing video
Love it